Solubility product of silver bromide is 5times 10^{-13} . Thequantity of potassium bromide (molar mass taken 120 gmol^{-l}) to be added to 1 litre of 0.05 M solution ofsilver nitrate to start

SOLVED: Does empirical formula of precipitate solution solution B precipitate form when A and B are mixed? ammonium nitrate sodium acetate yes silver nitrate potassium bromide yes no manganese(II) bromide zinc nitrate

Solubility product of silver bromide is `5.0xx10^(-13)`. The quantity of potassium bromide - YouTube
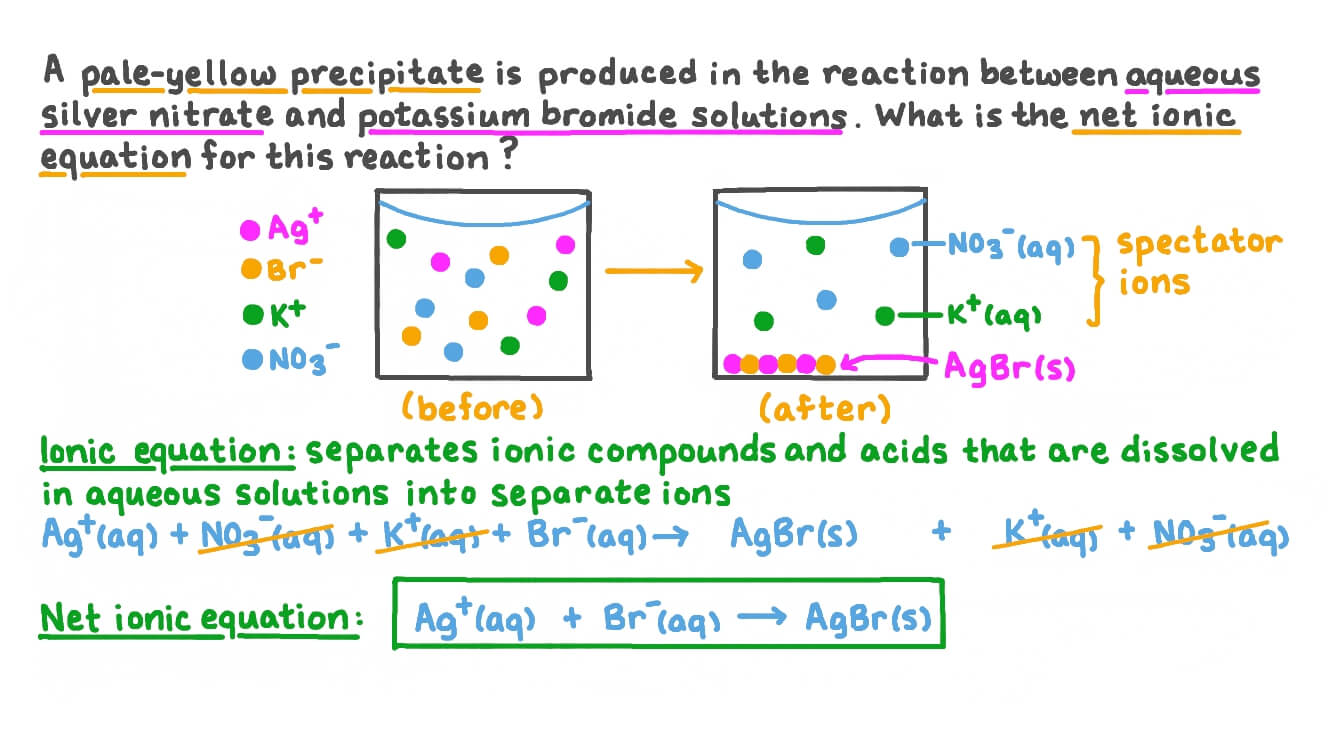
Question Video: Writing a Net Ionic Equation for the Reaction between Silver Nitrate and Potassium Bromide Solutions | Nagwa

Solubility product of silver bromide is 5.0times 10^{-13}The quantity of potassium bromide (molar masstaken as 120 g mol^{-1}) to be added to 1 litre of 0.05 Msolution of silver nitrate to start