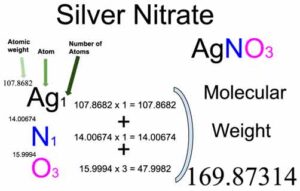
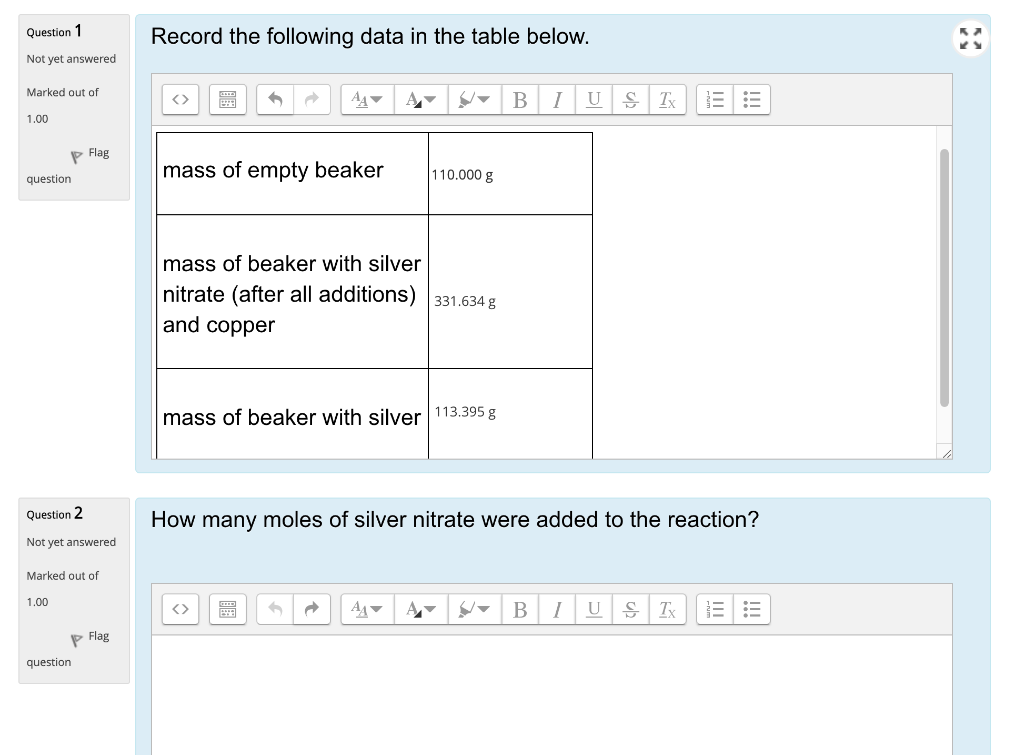
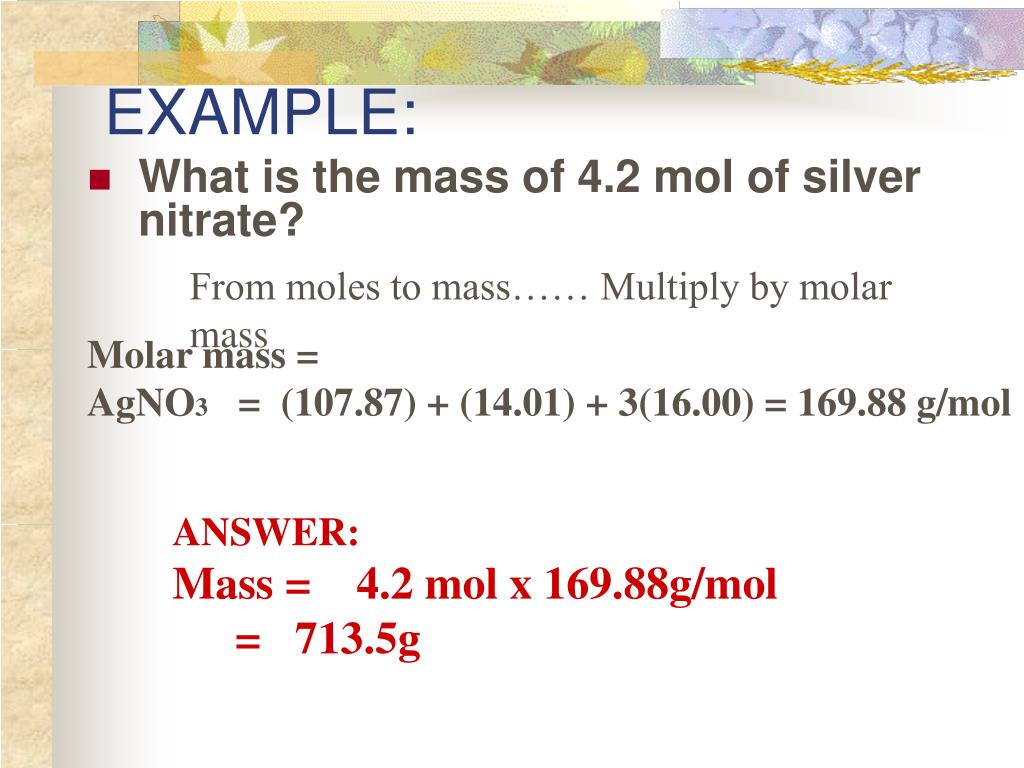SOLVED: Silver nitrate AgNO3 (molar mass = 169.9 g/mol), reacts with sodium chromate Na2CrO4 (molar mass = 161.9 g/mol), to from silver chromate (molar mass =331.7 g/mol) and sodium nitrate. If 45.5mL

The value of the observed and calculated molecular weight of silver nitrate is 92.64 and 170, respectively. The degree of dissociation of silver nitrate is:

The values of observed and calculated molecular mass of Ca(NO3)2 are 65.4 and 164 respectively. The degree of ionisation of the salt will be:

SOLVED: Stoichiometry What mass of silver chloride, AgCl; precipitates if 5.00 g of silver nitrate, AgNO3,is added t0 a sodium chloride solution? AgNO3 NaCl AgCl NaNO3 If0.162 g of aluminum is burned

SOLVED: Question 5 (1 point) NaCl(aq) AgNO3(aq) AgCl(s) NaNO3(aq) molar masSNaCl 58.43 g/mol molar massAgNO3 "169.83 g/mol molar massAgci 143.31 g/mol molar masSNaNO3 84.95 g/mol If 50. grams of sodium chloride is
The value of observed and calculated molecular weights of silver nitrate are 92.64 and 170 respectively. The degree of dissociation of silver nitrate is

The value of the observed and calculated molecular weight of silver nitrate is 92.64 and 170, respectively. The degree of dissociation of silver nitrate is:

Question Video: Calculating the Mass of Solute Needed to Prepare a Solution with a Desired Concentration and Volume | Nagwa