Nonradical Zinc−Barbier Reaction for Diastereoselective Synthesis of Vicinal Amino Alcohols | Journal of the American Chemical Society

The Barbier–Grignard-type arylation of aldehydes using unactivated aryl iodides in water | Nature Communications
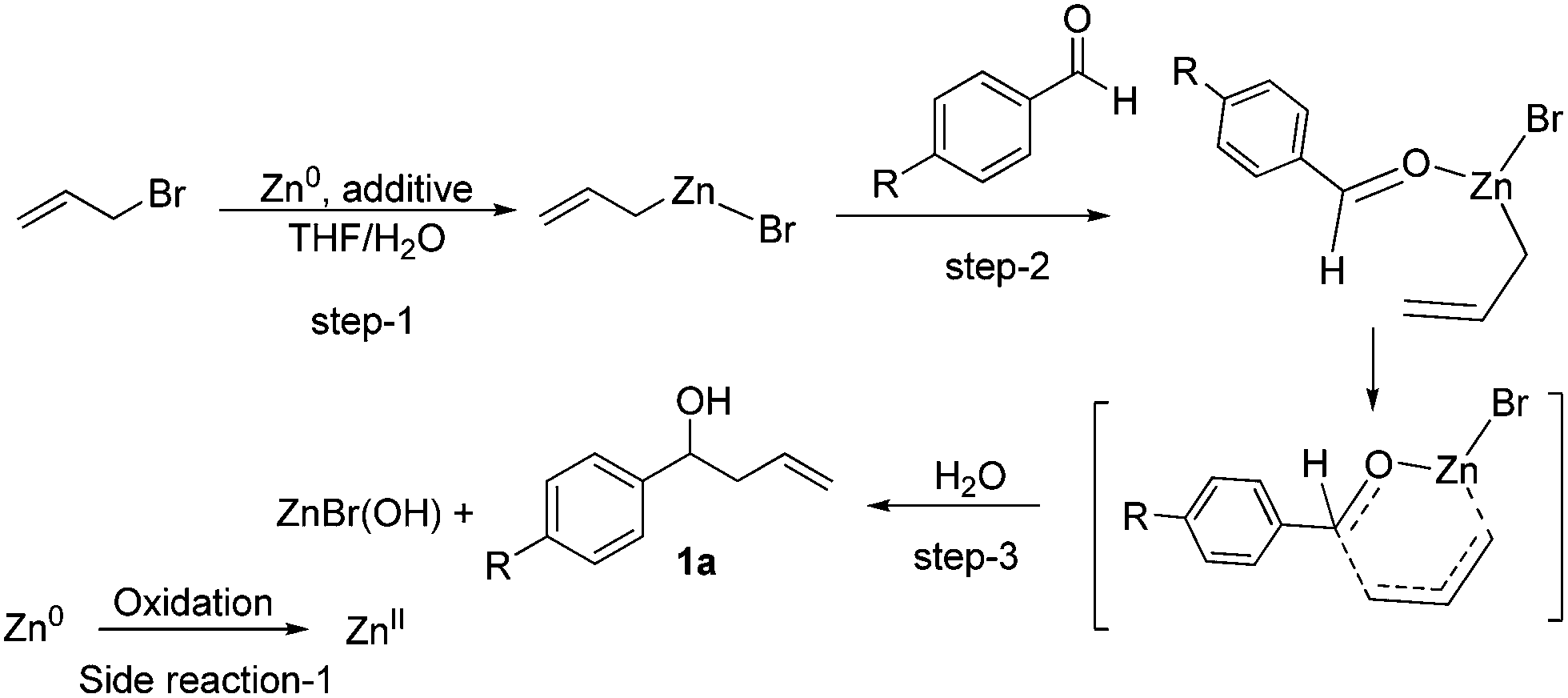
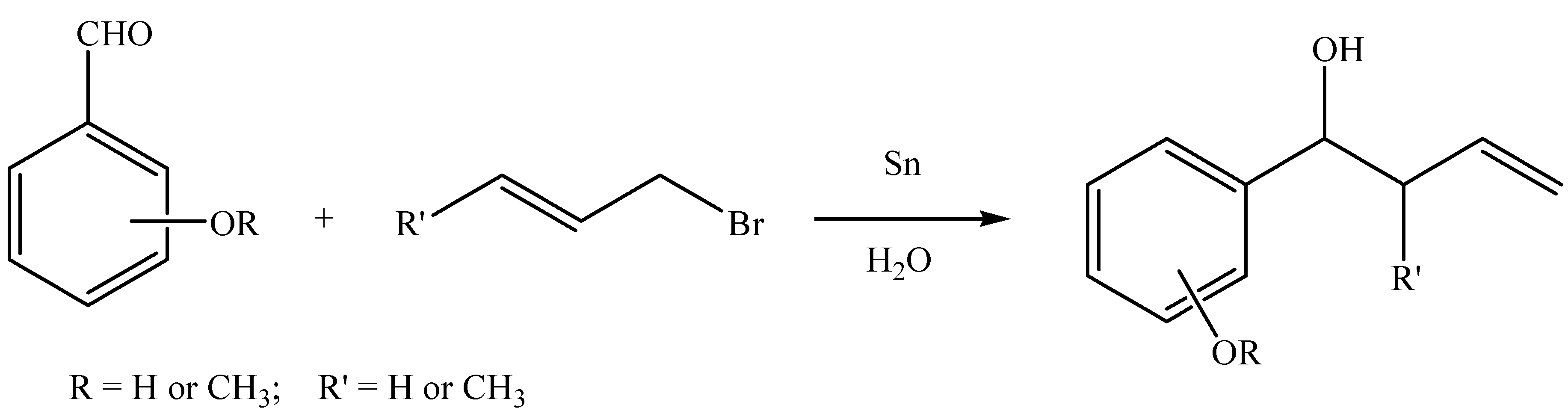
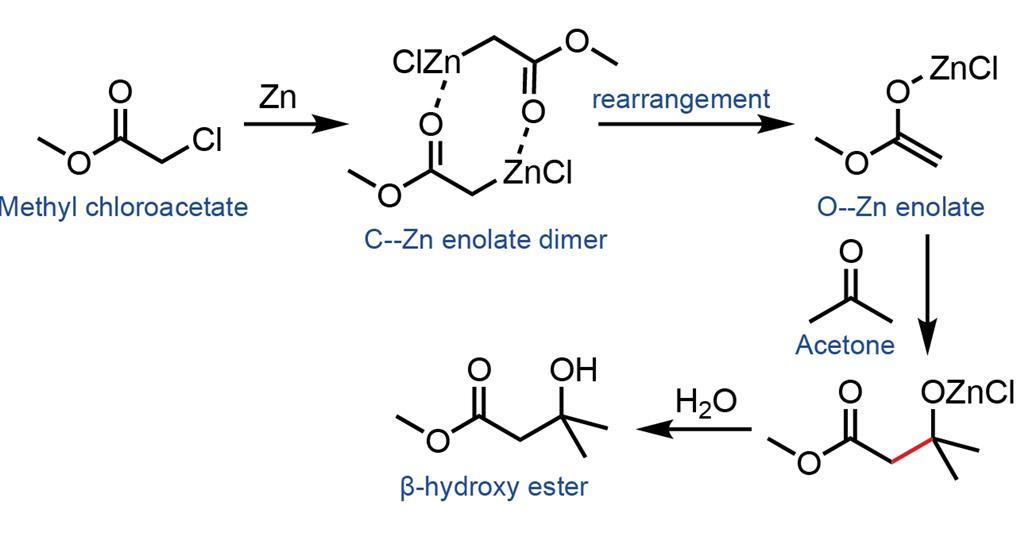
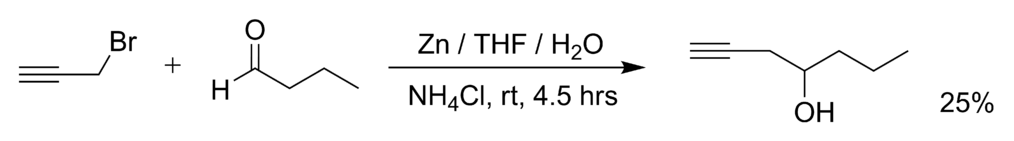
Waste management in zinc promoted allylation of aldehyde - New Journal of Chemistry (RSC Publishing) DOI:10.1039/C5NJ03569D
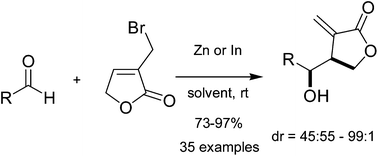
Zinc or indium-mediated Barbier-type allylation of aldehydes with 3-bromomethyl-5H-furan-2-one in aqueous media: an efficient synthesis method for α-methylene-γ-butyrolactone - Organic & Biomolecular Chemistry (RSC Publishing)
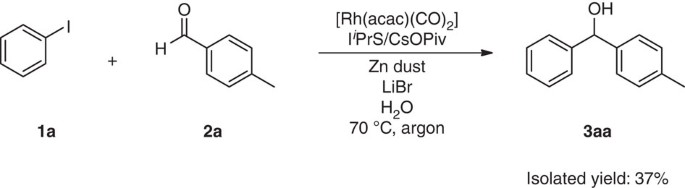
The Barbier–Grignard-type arylation of aldehydes using unactivated aryl iodides in water | Nature Communications
Photocatalytic Barbier reaction – visible-light induced allylation and benzylation of aldehydes and ketones
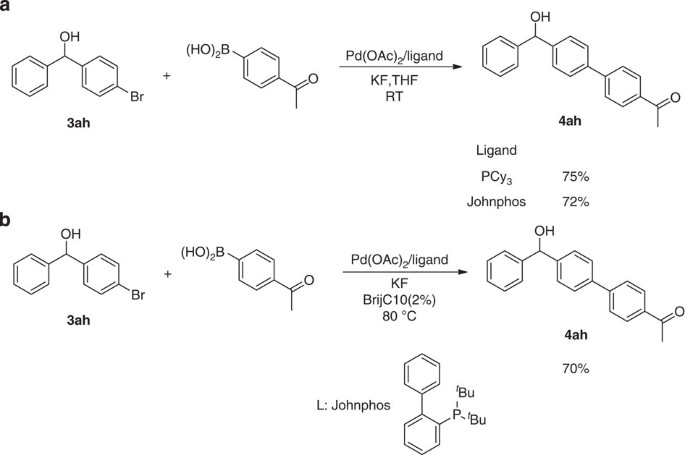
The Barbier–Grignard-type arylation of aldehydes using unactivated aryl iodides in water | Nature Communications